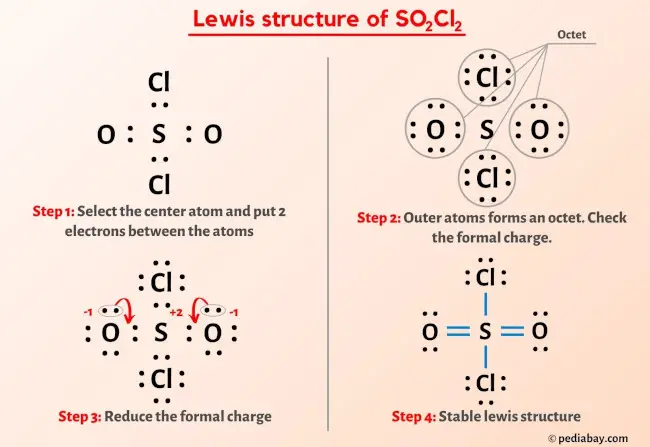
Sulphuryl chloride SO2Cl2, reacts with H2O to give mixture of H2SO4 and HCl. Aqueous solution of 1mole of SO2Cl2 will be neutralized by:a)3 moles of NaOHb)2 moles of Ca(OH)2c)Bothd)None of these Correct
16. So2cl2 reacts with H2o gives H2so4 and Hcl determine volume of 0.2M Ba(oH)2 required to neutralised 25ml of 0.1 M so2cl2

SOLVED: Given the data in the table below and ΔH°rxn for the reaction below, SO2Cl2 (g) + 2 H2O (l) â†' H2SO4 (l) + 2 HCl (g) ΔH° = -118 kJ/mol, what

SOLVED: Use the ΔH°f information provided to calculate ΔH°rxn for the following: ΔH°f (kJ/mol) SO2Cl2(g) + 2 H2O(l) â†' 2 HCl(g) + H2SO4(l) ΔH°rxn = ? SO2Cl2(g) -364 H2O(l) -286 HCl(g) -92

53. Sul CU (3) CIO Sulphuryl chloride SO.CI, reacts with H,O to (4) MnO give mixture of H.SO, and HCL. Aq. solution 1 of 1 mol so.Cl, will be neutralised by: (1)
SO2Cl2 on reaction with excess of water results into acidic mixture SO2Cl2 + 2H2O → H2SO4 + 2HCl - Sarthaks eConnect | Largest Online Education Community

SOLVED: 22. Given the data in the table below and ΔH^∘rxn for the reaction SO2Cl2( g)+2 H2O(l) →H2SO4(l)+2 HCl(g) ΔH^∘=-62 kJ ΔH^∘f of HCl(g) is kJ / mol. A. -184 B. 60
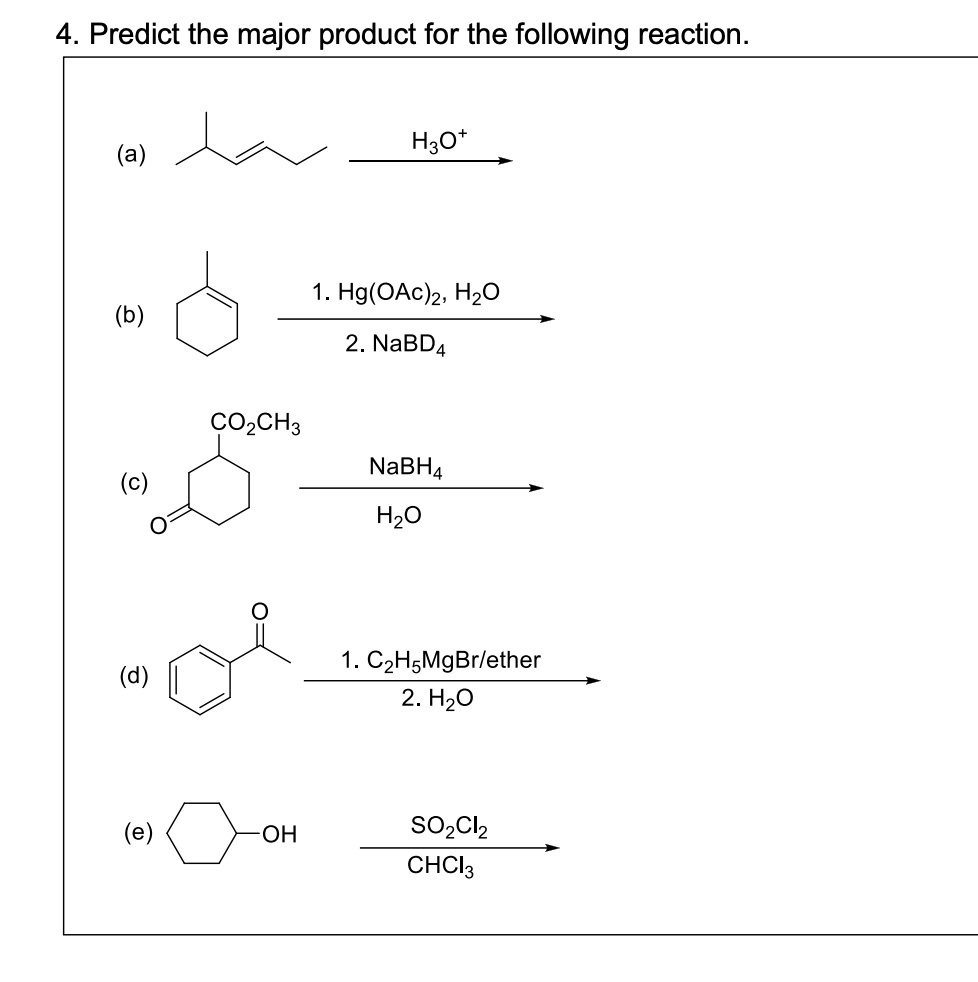
SOLVED: 4. Predict the major product for the following reaction H3O a 1.Hg(OAc)2,H2O (b) 2.NaBD4 CO2CH3 NaBH4 H2O 1.C2H5MgBr/ether 2.H2O SO2Cl2 CHCI3 (e) OH
13.Sulphuryl chloride, SO2Cl2, reacts with H2O to give mixture of H2So4 and HCL. Aqueous solution of 1 mol SO2Cl2 will be neutralised by ?. (1) 3 moles of NaOH (3) Both (1)

Question Benzoyl chloride is prepared from benzoic acid by: A. Cl2, hv B. SO2Cl2 C. SOCI2 D. Cl2, H2O