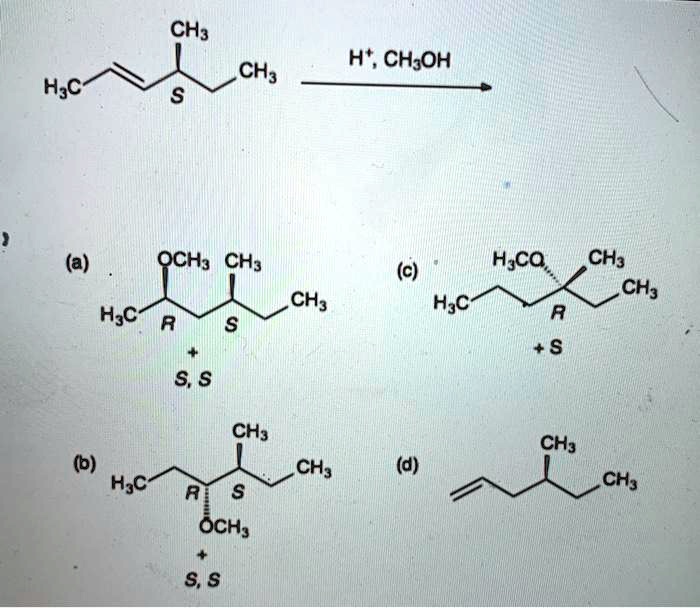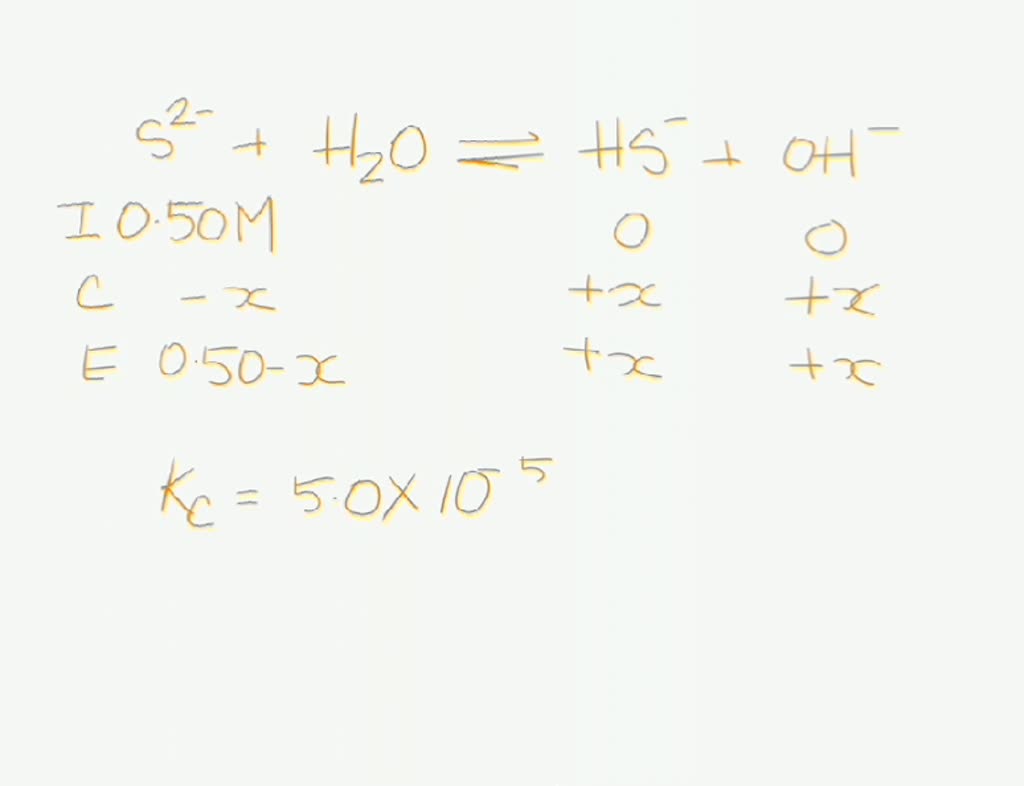
SOLVED: The sulfide ion, S2-, reacts with water as a weak base: S2-(aq) + H2O â†' SH(aq) + OH-(aq) with an equilibrium constant of 5.0x10^-5. If sodium sulfide were dissolved in water

SO2+H2O=H2SO3 balance the chemical equation @mydocumentary838. so2+h2o=h2so3 balance equation. - YouTube

U2 complete the following SO2 + H+ + NO+ +? A SO2 + 2NO+ H2O + N2O+ so YOU MISSED B SO2 + 4NO+2H20 +2N20+5 C SO2 + 2NO+ 4H2O + N20+S4 D None of these

Effects of SO2 and H2O on low-temperature NO conversion over F-V2O5-WO3/TiO2 catalysts - ScienceDirect
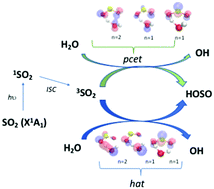
Triplet state promoted reaction of SO2 with H2O by competition between proton coupled electron transfer (pcet) and hydrogen atom transfer (hat) processes - Physical Chemistry Chemical Physics (RSC Publishing)

Predicting the Formation of Sulfur-Based Brønsted Acids from the Reactions of SOx with H2O and H2S | The Journal of Physical Chemistry A

SO2, H2O, SO3, and atomic oxygen mixing ratios at 80 km vary with SO2... | Download Scientific Diagram
if S + O2— > SO2 ; ( DH= 298.2 KJ ) SO2 +1/2O2——>SO3 ( DH= 98.7 KJ) SO3 + H2O——>H2SO4 (DH= 130.2 KJ) H2 + 1/2H2O—–> H2O ( DH= 287.3KJ) then
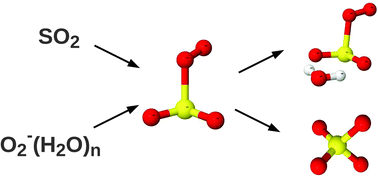
On the gas-phase reaction between SO2 and O2−(H2O)0–3 clusters – an ab initio study - Physical Chemistry Chemical Physics (RSC Publishing)

School2Office - You all have heard of H2O but here is a new formula "S2O" for you. Start running the gears of your brain and keep asking what is S2O? Stay tuned

Simulated infrared spectra of SO2, SO3 and H2O calculated at T = 600 K... | Download Scientific Diagram