Jornal Médico - Adempas (riociguat) da Bayer aprovado na UE para o tratamento de duas formas graves de hipertensão pulmonar
Адемпас (Adempas) риоцигуат (riociguat) 0.5 мг, 1 мг, 1.5 мг, 2 мг, 2.5 мг таб. Европа недорого купить в Казахстане с доставкой | med-pharm

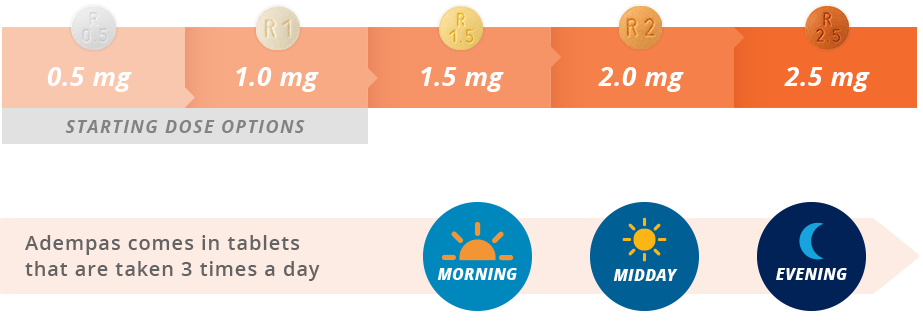
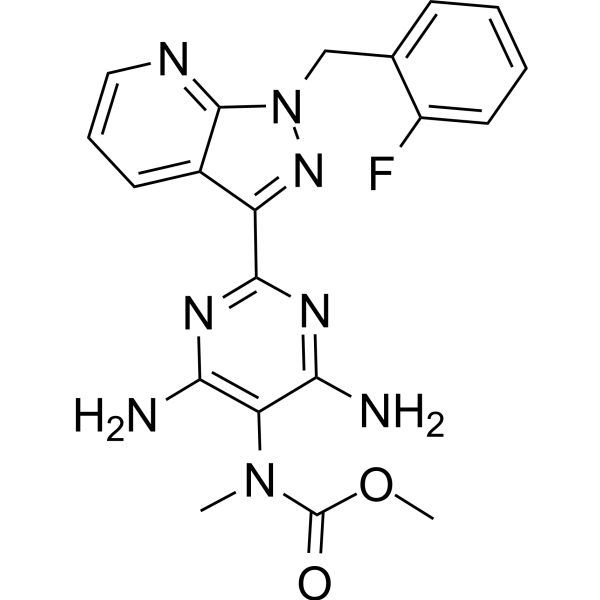
The U.S. Food and Drug Administration approved Adempas (riociguat) to treat adults with two forms of pulmonary hypertension. | New Drug Approvals