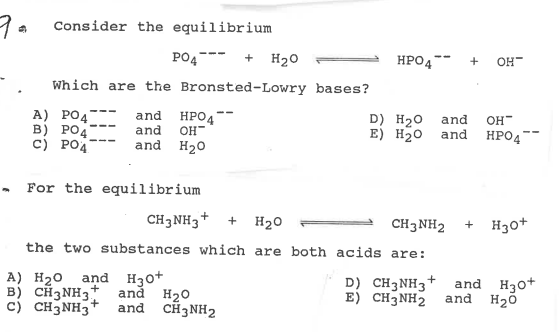
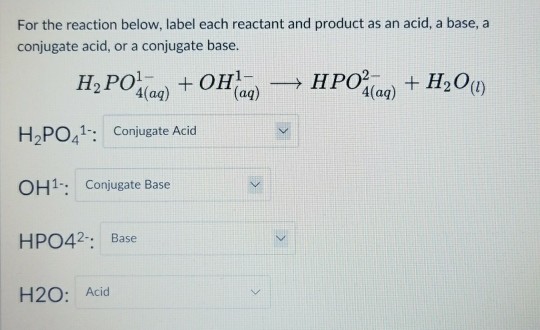
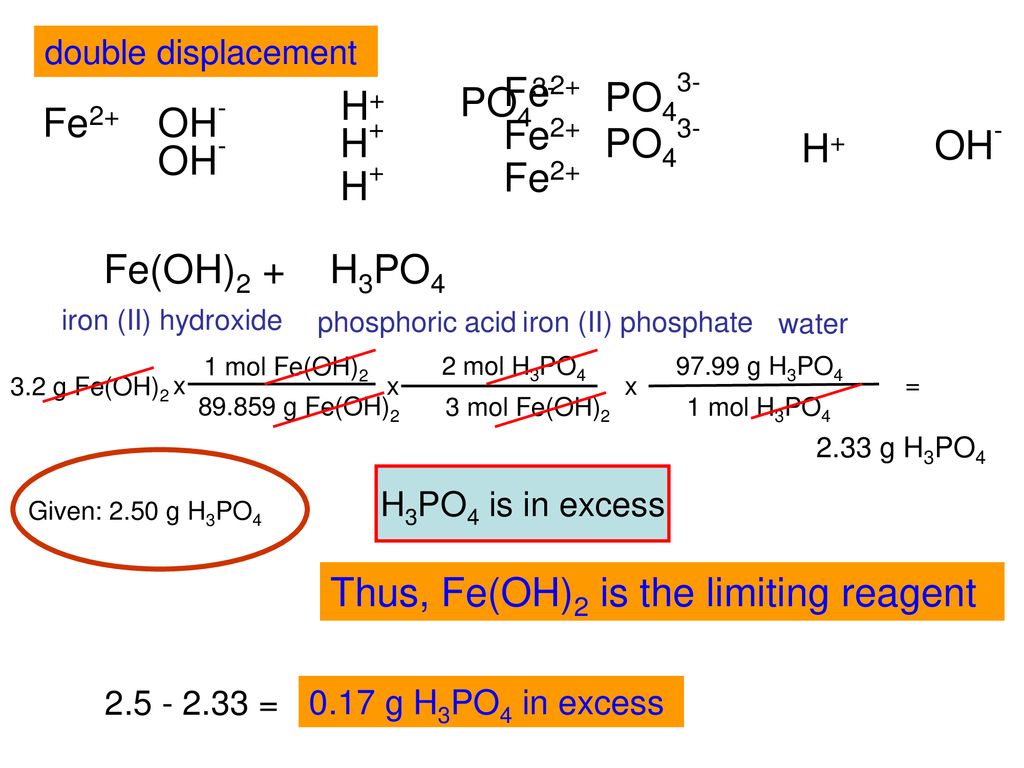
42. Three reactions involving H,PO, are given below 1. H,PO4 + H20 - H30* + H2PO4 II. H POE +H20- HPO2 + H20+ III. HỌPO4 + OH - H3PO4 +02- In which

SOLVED: 1. Balance the equation H3PO4 + Ca(OH)2 —> Ca3(PO4)2 + H2O and show the tally table. 2. Draw a colored particle diagram of your total balanced chemical equation. Show a key.

SOLVED: When the equation Ca(OH)2 + H3PO4 –> Ca3(PO4)2 + H2O is balanced, the coefficient for H2O is

The experimental X-Ray diffraction pattern of Fe3(PO4)2.8H2O, allowed... | Download Scientific Diagram

Figure 1 from Electrochemical analysis of Co3(PO4)2·4H2O/graphene foam composite for enhanced capacity and long cycle life hybrid asymmetric capacitors | Semantic Scholar

Ca(OH)2 +H3PO4 =Ca3(PO4)2+ H2O Balanced Equation|Cacium Hydroxide +Phosphoric Acid Balanced Equation - YouTube
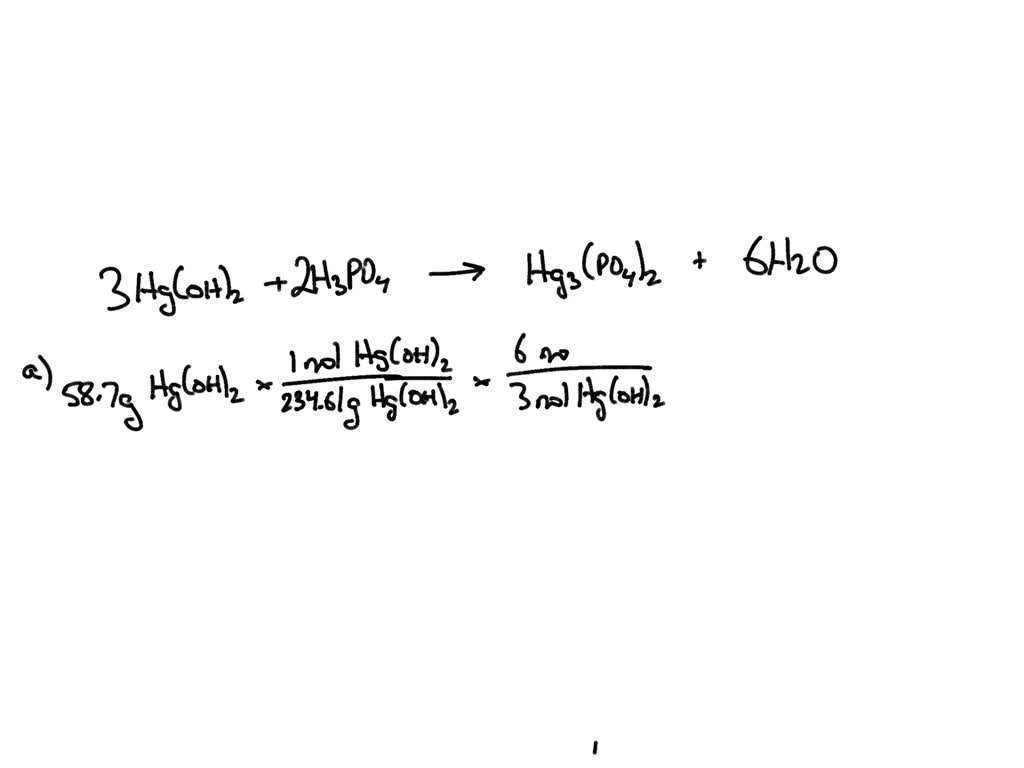
SOLVED: Use the reaction to solve the following problems. Hg(OH)2 + H3PO4→ Hg3(PO4)2 + H2O A) Given 58.7 grams of Hg(OH)2, how many moles of H2O will be produced? If you react
Magnetic susceptibility and crystal structure of the new oxovanadium phosphate (NH4)Zn(H2O)(VO)2(PO4)2(H2PO4)

Synthesis and Characterization of a Novel Hydrated Layered Vanadium(III) Phosphate Phase K3V3(PO4)4·H2O: A Functional Cathode Material for Potassium-Ion Batteries | ACS Omega