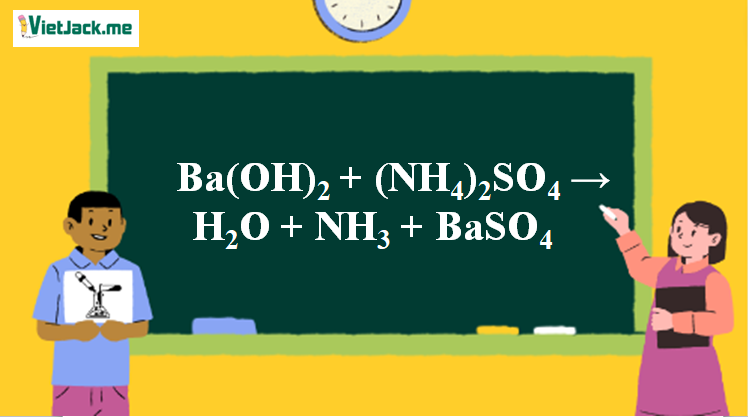SOLVED: The phase diagram for the ternary system NH4Cl / (NH4)2SO4 H2O at 25°C is shown below: 0.2 0.8 P=V 0.4 0.6 P=2 0.6 P=2 0.4 P=3 0.8 0.2 NH4Cl (NH4)2SO4 0.2

Images of frozen (NH4)2SO4/H2O films. a, b, Images taken from the same... | Download Scientific Diagram
![Ammonium Sulfate [(NH4)2SO4] - Structure, Molecular mass, Preparation, Properties, Uses and FAQs of Ammonium Sulfate. Ammonium Sulfate [(NH4)2SO4] - Structure, Molecular mass, Preparation, Properties, Uses and FAQs of Ammonium Sulfate.](https://cdn1.byjus.com/wp-content/uploads/2018/09/ammonium-sulfate-structure-700x239.png)
Ammonium Sulfate [(NH4)2SO4] - Structure, Molecular mass, Preparation, Properties, Uses and FAQs of Ammonium Sulfate.

Phase Diagram for the Na2SO4–(NH4)2SO4–MEA–H2O System at Elevated Temperature | Journal of Chemical & Engineering Data

Acid + HNO3 + ....... + Base ....... 2NH,OH salt KNO3 + + Water H2O - - - - (NH4)2SO4 + ........ + KOH KBr +







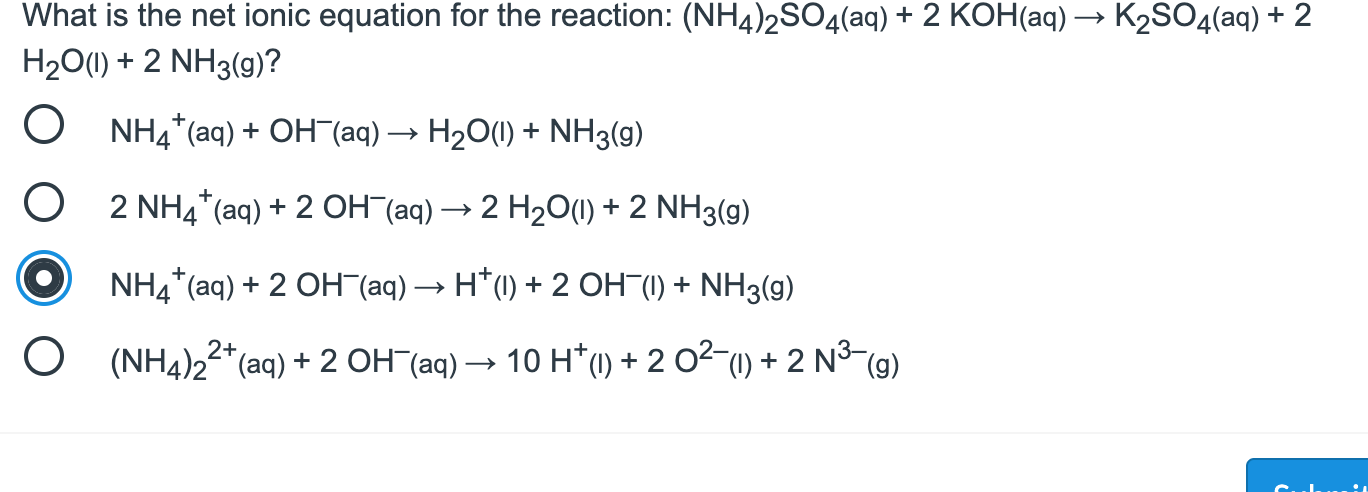
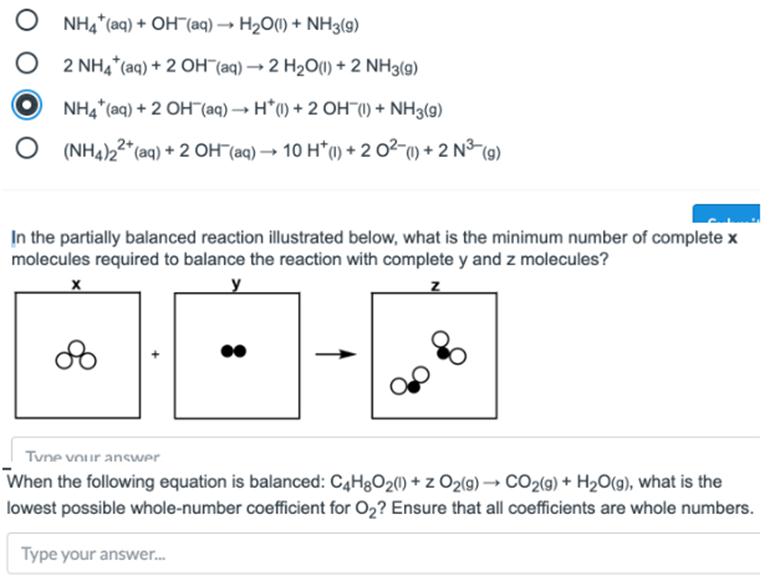


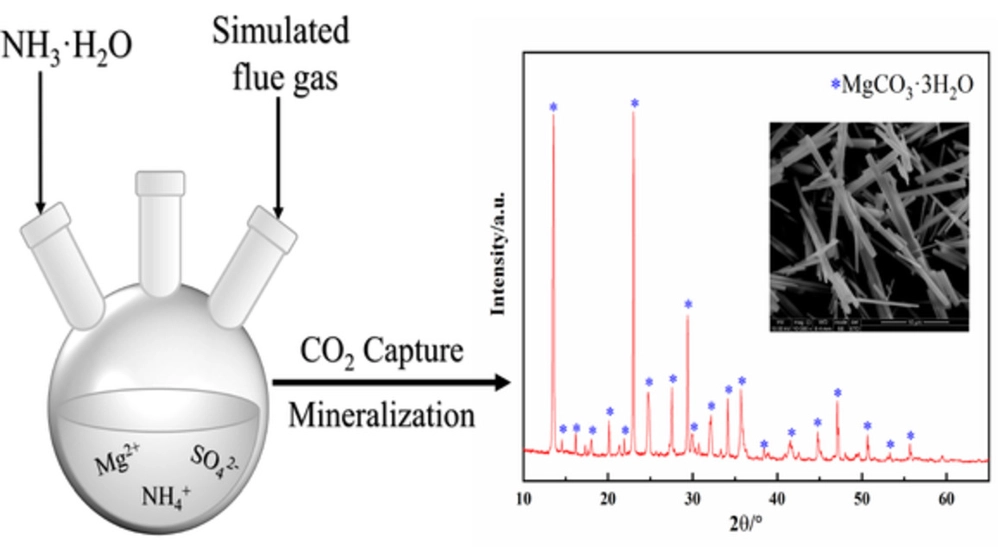
2SO4.jpg)