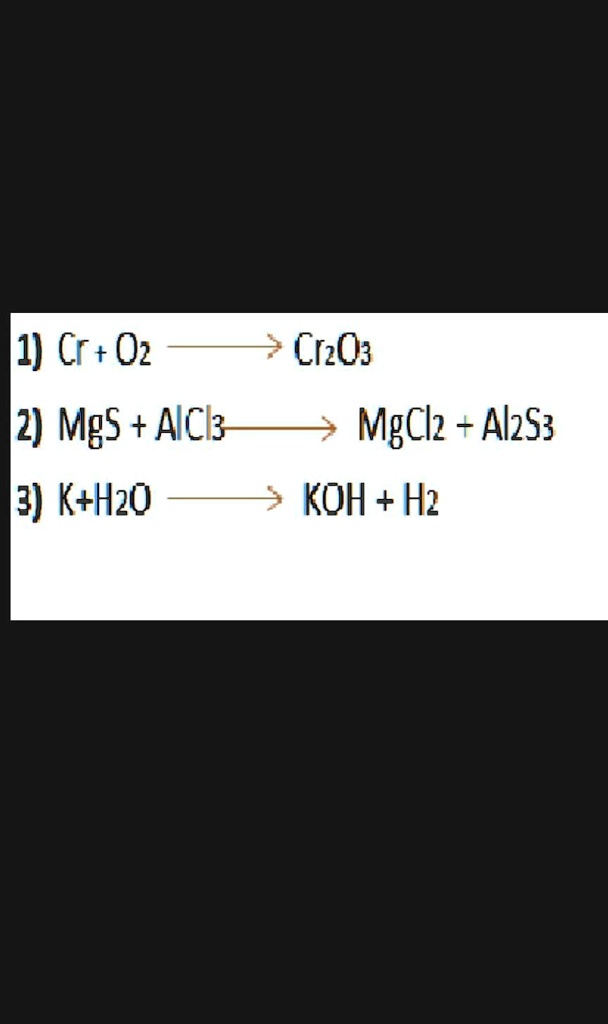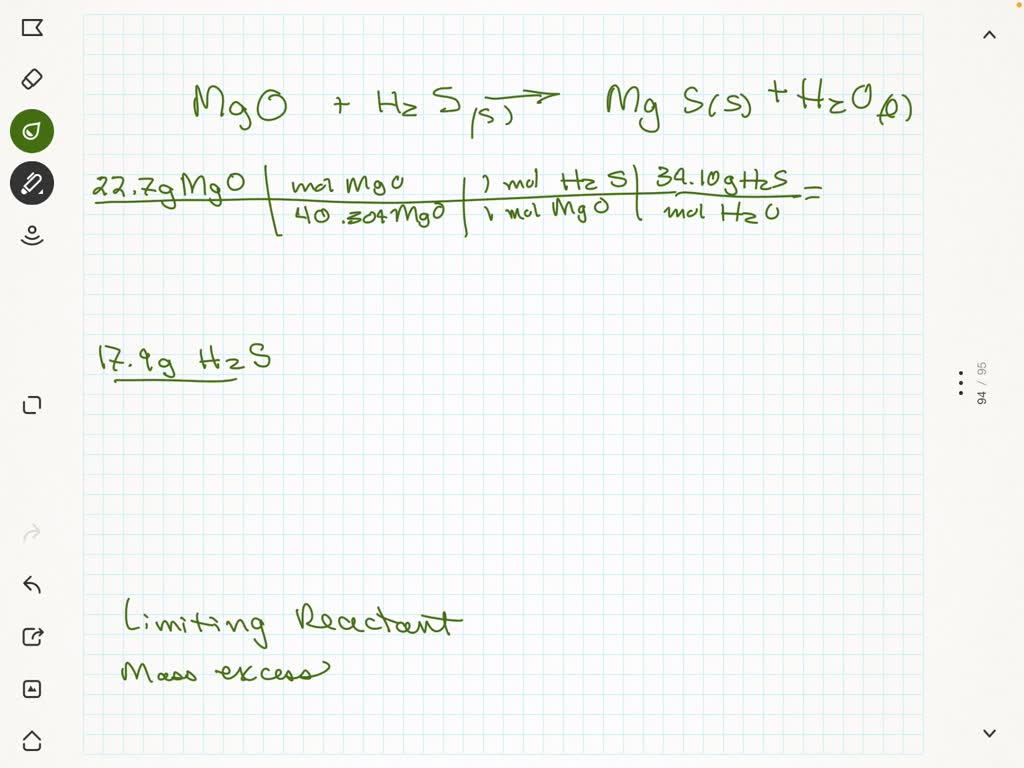
SOLVED: Given the initial amounts listed, what is the limiting reagent, and what is the mass of the leftover reagent? 22.7 g of MgO(s) + 17.9 g of H2S —> MgS(s) +

SOLVED: Using the "hard-soft" acid-base concept, predict the direction of the following reactions. (2 pts each) CaS + 2 H2O â†' Ca(OH)2 + H2S BaO + MgS â†' BaS + MgO CH3Hg(H2O) +

Sustainability | Free Full-Text | Enhanced Effect of Metal Sulfide Doping ( MgS-TiO2) Nanostructure Catalyst on Photocatalytic Reduction of CO2 to Methanol

Spatial variation of DOC (MGS, mountain gorge section; UPS, urban plain... | Download Scientific Diagram

SOLVED: The net ionic equation for the reaction of solutions containing sodium sulfide and magnesium nitrate will include which of the following: MgS (s) Mg2+ (aq) 2 NO3- (aq) H2O (l) S2- (
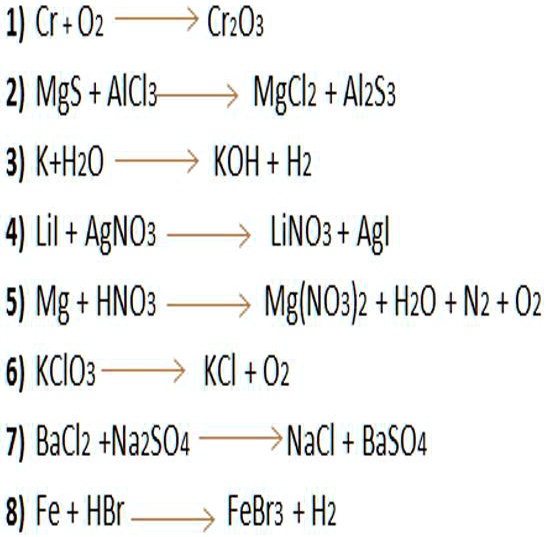
SOLVED: balancear por el método del tanteo 1) Cr+02 Cr203 2) MgS + Alcl3 Mgcl2 4 Al2s3 3) K+H2o KOH + H2 4) Lil + AgNO3 LiNo3 + Ag| 5) Mg 1

Calcium oxide and magnesium sulfide each react with acid. I CaO(s) + 2H*(aq) → Ca²+ (aq) + H₂O(1) MgS(s) + - Brainly.in








![Telugu] What product will be obtained when magnesite is dissolved in Telugu] What product will be obtained when magnesite is dissolved in](https://static.doubtnut.com/ss/web-overlay-thumb/7112675.webp)