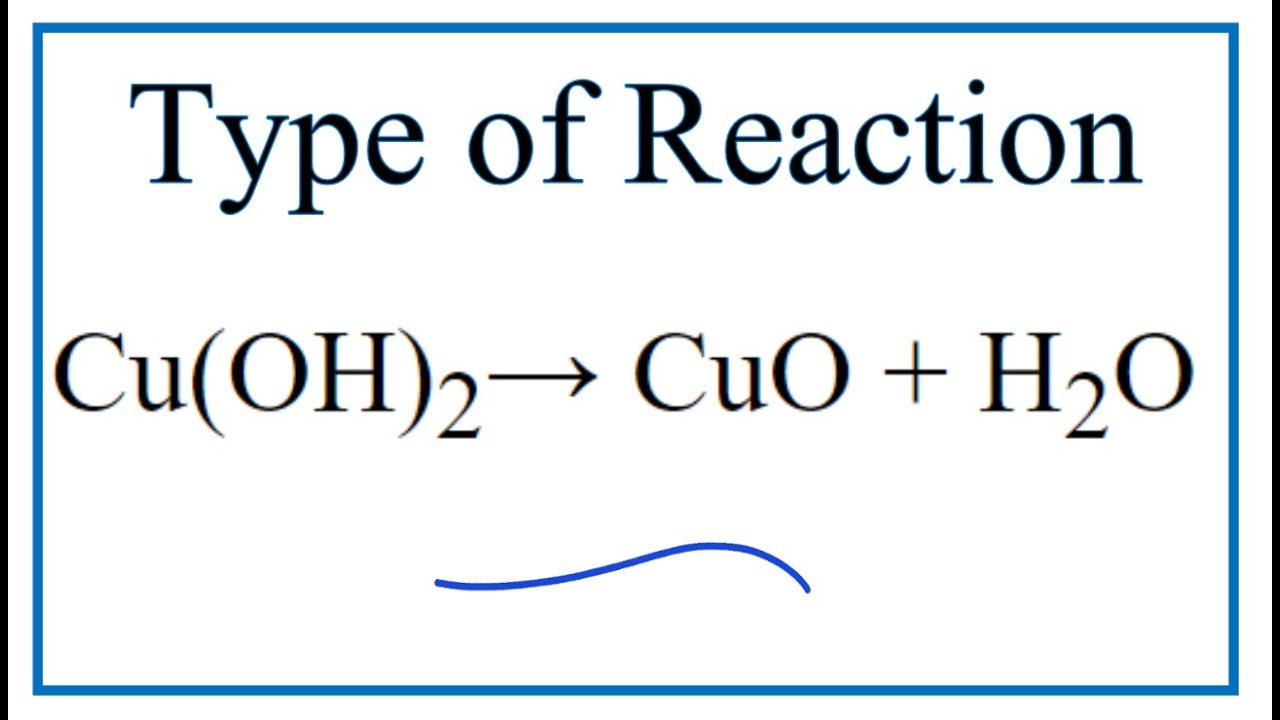
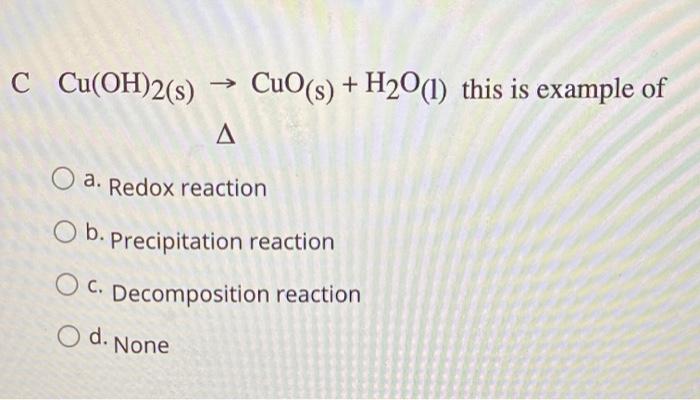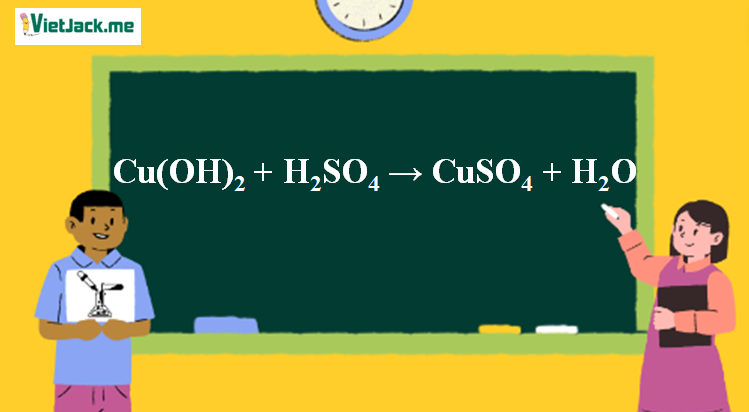Copper hydroxide -Cu(OH)2 - Structure, Molecular Mass, Physical Properties, Chemical Properties, Uses and FAQs of Copper hydroxide.
Crystal structure of Cu(OH) 2 (a) and a proposed structure model of... | Download Scientific Diagram

Figure 2 from Simple Template-Free Solution Route for the Controlled Synthesis of Cu(OH)2 and CuO Nanostructures | Semantic Scholar


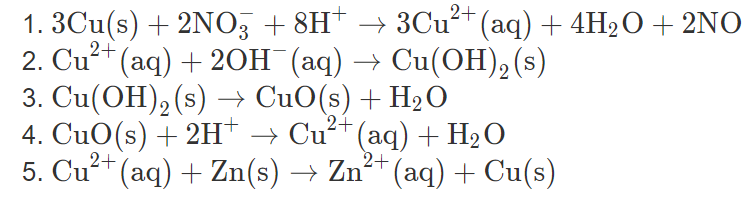








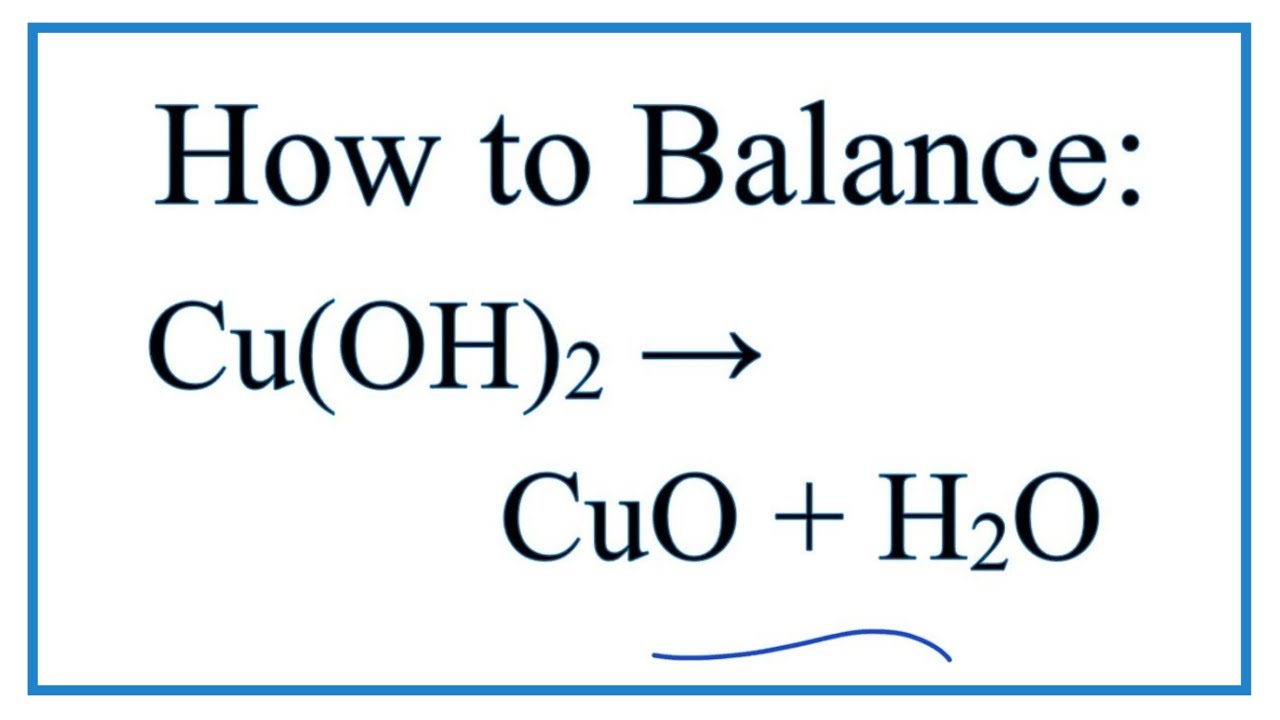

![Cu(OH2)6]2+ - Copper(II) hydroxide Cu(OH2)6]2+ - Copper(II) hydroxide](https://www.chemtube3d.com/images/gallery/inorganicsjpgs/cuoh262_.jpg)