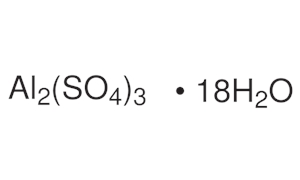
ALUMINIUM SULPHATE practical iron free Extra Pure | Lab chemical distributors, Lab chemicals exporter, Laboratory chemical suppliers, Lab chemical supplier, Laboratory Chemicals, Laboratory chemicals manufacturer, Lab chemical manufacturer, Alpha ...

High Quality Chemical Formula Al2 So4 3.18H2O Aluminum Sulfate of Aluminum - China Aluminium Sulfate, Aluminium Sulfate 17% Min | Made-in-China.com
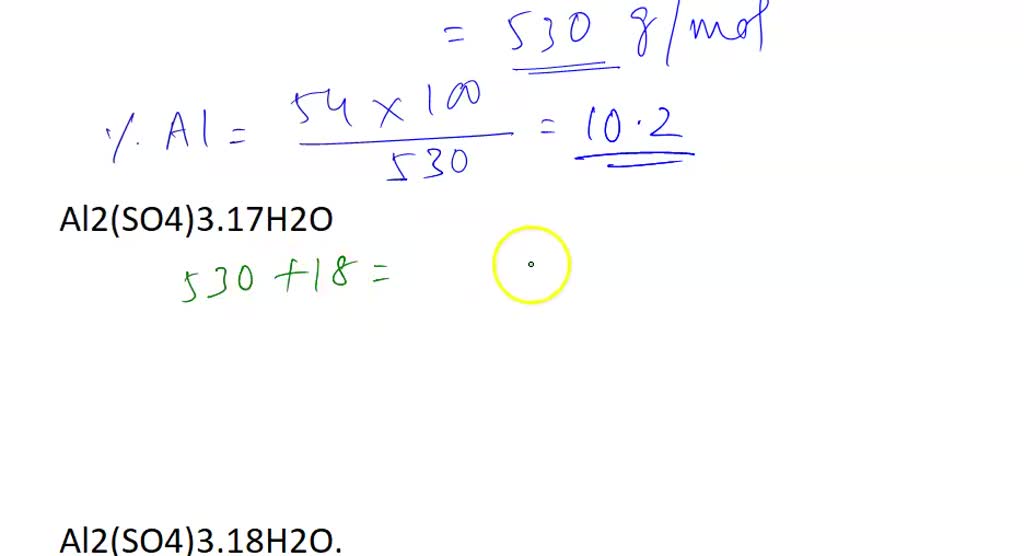
SOLVED: Aluminum sulfate occurs as a hydrate, commonly with 16-18 moles of H2O. Calculate the molar masses and Al% for each Al2(SO4)3*16H2O, Al2(SO4)3*17H2O, and Al2(SO4)3*18H2O.

Al2(SO4)3.18H2O after calcium for 2 h at 500-1300ºC with the weight of... | Download Scientific Diagram

Aluminium sulphate solution 0.2 % Al2(SO4)3 * 18 H2O for blue number determination Contents:, 48,91 €

Jual Aluminium (III) Sulfat, 18-hidrat | Al2(SO4)3.18H2O 500 gram - Kota Bandung - Rofa Laboratorium Centre | Tokopedia












![Aluminium Sulfate Octahydrate [Al2(SO4)3.8H2O] Molecular Weight Calculation - Laboratory Notes Aluminium Sulfate Octahydrate [Al2(SO4)3.8H2O] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2023/03/aluminium-sulfate-octahydrate-molecular-weight-calculation-300x178.jpg)



